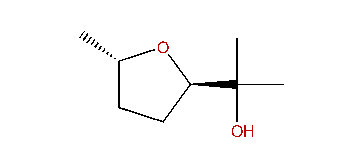
| (2R,5S)-2-(1-Hydroxy-1-methylethyl)-5-methyltetrahydrofuran |
| Formula: | C8H16O2 |
| CAS#: | 3284-88-6 |
| MW: | 144.21 |
[MS]
[ NMR ]
[Behavioural function]
[Chemdraw]
|
|
Reference(s) for synthesis of (2R,5S)-2-(1-Hydroxy-1-methylethyl)-5-methyltetrahydrofuran
| Ishihara, K., Mori, A., and Yamamoto, H. 1987. Stereoselective reduction of bicyclic acetals. A method for reductive generation of heterocyclic ring systems. Tetrahedron Lett. 28:6613-6616. |
| |
| Ishihara, K., Mori, A., and Yamamoto, H. 1990. Stereoselective reduction of acetals. A method for reductive generation of heterocyclic ring systems. Tetrahedron. 46:4595-4612. |
| |
| Kennedy, R.M., and Tang, S. 1992. Directed oxidative cyclization of 5-hydroxyalkenes with rhenium oxide. Tetrahedron Lett. 33:3729-3731. |
| |
| Lattanzi, A., Sala, G.D., Russo, M., and Scettri, A. 2001. Diastereoselective titanocene-catalyzed oxidative cyclization of bishomoallylic alcohols. Syn. Lett. 9:1479-1481. |
| |
| Steinreiber, A., Edegger, K., Mayer, S.F., and Faber, K. 2001. Enantio- and diastereo-convergent synthesis of (2R,5R)- and (2R,5S)-Pityol through enzyme-triggered ring closure. Tetrahedron: Asymmetry. 12:2067-2071. |
| |
| Tang, S., and Kennedy, R.M. 1992. Stereocontrolled oxidation of 5-hydroxyalkenes with rhenium oxide. Tetrahedron Lett. 33:5299-5302. |
| |
|







